Page 99 - lef-105
P. 99
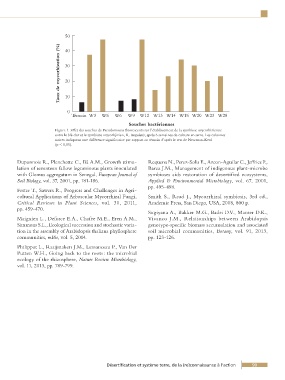
50
Taux de mycorhization (%) 30
40
20
10
0
Témoin W3 W5 W6 W9 W12 W13 W14 W15 W20 W23 W28
Souches bactériennes
Figure 1. Effet des souches de Pseudomonas fluorescents sur l’établissement de la symbiose mycorhizienne
entre le blé dur et le symbiote mycorhjizien, R. irregularis, après 6 semaines de culture en serre. Les colonnes
noires indiquent une différence significative par rapport au témoin d’après le test de Newman-Keul
(p < 0,05).
Duponnois R., Plenchette C., Bâ A.M., Growth stimu- Requena N., Perez-Solis E., Azcon-Aguilar C., Jeffries P.,
lation of seventeen fallow leguminous plants inoculated Barea J.M., Management of indigenous plant-microbe
with Glomus aggregatum in Senegal, European Journal of symbioses aids restoration of desertified ecosystems,
soil Biology, vol. 37, 2001, pp. 181-186. Applied & Environmental microbiology, vol. 67, 2001,
Fester T., Sawers R., Progress and Challenges in Agri- pp. 495-498.
cultural Applications of Arbuscular Mycorrhizal Fungi, Smith S., Read J., Mycorrhizal symbiosis, 3rd ed.,
Critical Reviews in Plant sciences, vol. 30, 2011, Academic Press, San Diego, USA, 2008, 800 p.
pp. 459-470. Sugiyana A., Bakker M.G., Badri D.V., Manter D.K.,
Maignien L., Deforce E.A., Chafee M.E., Eren A.M., Vivanco J.M., Relationships between Arabidopsis
Simmons S.L., Ecological succession and stochastic varia - genotype-specific biomass accumulation and associated
tion in the assembly of Arabidopsis thaliana phyllosphere soil microbial communities, Botany, vol. 91, 2013,
communities, mBio, vol. 5, 2014. pp. 123-126.
Philippot L., Raaijmakers J.M., Lemanceau P., Van Der
Putten W.H., Going back to the roots : the microbial
ecology of the rhizosphere, Nature Review microbiology,
vol. 11, 2013, pp. 789-799.
Désertification et système terre, de la (re)connaissance à l’action 99
Taux de mycorhization (%) 30
40
20
10
0
Témoin W3 W5 W6 W9 W12 W13 W14 W15 W20 W23 W28
Souches bactériennes
Figure 1. Effet des souches de Pseudomonas fluorescents sur l’établissement de la symbiose mycorhizienne
entre le blé dur et le symbiote mycorhjizien, R. irregularis, après 6 semaines de culture en serre. Les colonnes
noires indiquent une différence significative par rapport au témoin d’après le test de Newman-Keul
(p < 0,05).
Duponnois R., Plenchette C., Bâ A.M., Growth stimu- Requena N., Perez-Solis E., Azcon-Aguilar C., Jeffries P.,
lation of seventeen fallow leguminous plants inoculated Barea J.M., Management of indigenous plant-microbe
with Glomus aggregatum in Senegal, European Journal of symbioses aids restoration of desertified ecosystems,
soil Biology, vol. 37, 2001, pp. 181-186. Applied & Environmental microbiology, vol. 67, 2001,
Fester T., Sawers R., Progress and Challenges in Agri- pp. 495-498.
cultural Applications of Arbuscular Mycorrhizal Fungi, Smith S., Read J., Mycorrhizal symbiosis, 3rd ed.,
Critical Reviews in Plant sciences, vol. 30, 2011, Academic Press, San Diego, USA, 2008, 800 p.
pp. 459-470. Sugiyana A., Bakker M.G., Badri D.V., Manter D.K.,
Maignien L., Deforce E.A., Chafee M.E., Eren A.M., Vivanco J.M., Relationships between Arabidopsis
Simmons S.L., Ecological succession and stochastic varia - genotype-specific biomass accumulation and associated
tion in the assembly of Arabidopsis thaliana phyllosphere soil microbial communities, Botany, vol. 91, 2013,
communities, mBio, vol. 5, 2014. pp. 123-126.
Philippot L., Raaijmakers J.M., Lemanceau P., Van Der
Putten W.H., Going back to the roots : the microbial
ecology of the rhizosphere, Nature Review microbiology,
vol. 11, 2013, pp. 789-799.
Désertification et système terre, de la (re)connaissance à l’action 99